|
Ergodicity describes the ability to predict the behavior of an ensemble from that of its
individual components. While non-ergodic processes are frequent in biological systems,
social behavior as well as economy, in chemistry they are usually restricted to small
molecules comprising only very few atoms. Accordingly, most chemical processes are
ergodic in that the result of a transformation of a reactant A into a product B is does not
depend on the way the transformation is achieved.
Using the strong infrared laser CLIO in France, an international team of chemists led by the
ERC grant holders Jana Roithová (Charles University, Prague) and
Detlef Schröder (Institute
of Organic Chemistry and Biochemistry, Prague) has now demonstrated that the restriction to
small molecules does not hold true for certain ion pairs containing S=O double bonds
(direct link to article in Angew. Chem. Int. Ed. ). In fact, in some of these systems
the non-ergodic behavior comprises a timescale sufficient for the formation of new chemical
bonds. In a nutshell, the origin of the non-ergodic effect is a hindered communication within
the ion pairs such that local overheating can occur.
The reviewers rated the contribution as a "Very Important Paper" (VIP) in Angewandte
Chemie, the leading journal of general chemistry; only about 2% of the contributions to
Angewandte Chemie are rated as VIP. Specifically, one reviewer commented that the
suggestion “goes against much of current dogma in molecular spectroscopy” and another
reviewer stated that “the hypothesis of non-ergodicity in such large systems… is fascinating”.
In addition to the relevance for the fundamental field of molecular science and spectroscopy,
the suggested local overheating might have possible relevance for related areas such as
heating with microwaves.
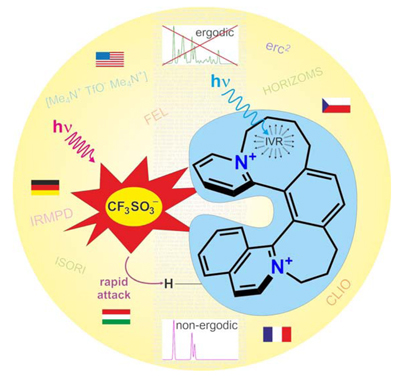
Illustration sketching the non-ergodic effects observed in non-covalent ion pairs.
Background info (downloadable pdf file)
For further information, please contact: detlef.schroeder@uochb.cas.cz
|