Maternal rhythmic behavior supports the development of the fetal biological clock (27.5. 2022)
NEW PUBLICATION
Before the fetal internal biological clock in the suprachiasmatic nuclei (SCN) of the hypothalamus begins to tick, the rhythmic behavior of the mother affects the function and development of this structure. This was found in a new study published by the team of Alena Sumová from the Institute of Physiology of the Academy of Sciences of the Czech Republic on May 24 in the journal PLOS Biology. This discovery contributes significantly to the understanding of the development of the internal clock and may find application in the treatment of premature babies.
The SCN are the central clock in our body, driven by the rhythmic switching on and off of the clock genes. The rhythmic activity of clock genes in SCN cells controls the activity of many other genes locally and elsewhere in the body, which ultimately affects a wide range of circadian rhythms in behavior, including food intake and sleep. However, this autonomous rhythmic gene activity of the SCN begins relatively late in fetal development, which raises the question of whether maternal signals may affect the gene activity in the SCN before it develops.
To investigate this question, the authors compared the daily gene activity profiles in SCN from fetuses developing in pregnant rats kept in the constant darkness under two different conditions. The control group of mothers had intact SCN and free access to food, while the other group of mothers had SCN surgically removed and had limited access to food for eight hours a day so that their locomotor activity showed a circadian rhythm even in the absence of a central clock. Using biostatistical analyzes they found that in the SCN of the fetuses of both groups, there was a small set of genes whose timing differed between the two groups, and a much larger set of genes whose activity oscillated in synchrony. Many of these genes could be assigned to two major neuronal processes, reflecting in the first case the ongoing development of the SCN and in the second case the earliest manifestation of their function. The data suggest that in the development of fetal SCN, maternal stimuli can replace the missing intercellular synaptic communication and control the rhythms of cell populations before the SCN clocks fully mature. The unexpected extent and specificity of SCN cell responses to maternal signals underlines the importance of a functioning maternal biological clock during pregnancy and points to the potential impact of the absence of such signals in preterm infants.

Greiner P., Houdek P., Sládek M., Sumová A.: Early rhythmicity in the fetal suprachiasmatic nuclei in response to maternal signals detected by omics approach. PLOS Biology; DOI, IF 8.029 Published: May 24, 2022
New findings on the structure of the FOXO4: p53 complex - a key factor in senescence regulation (22.4. 2022)
NEW PUBLICATION
Transcription factor p53 protects cells against tumorigenesis when subjected to various cellular stresses. Under stress conditions, p53 interacts with another transcription factor, FOXO4 (Forkhead box O 4), and together they increase the production of p21 protein, which triggers the process of cell aging (senescence). However, the molecular mechanism of upregulation of p21 transcription is still unclear. In the study published in the Protein Science journal, scientific teams of Dr. Obsilova (IPHYS CAS), prof. Obsil (Faculty of Science, Charles University and IPHYS CAS) and their colleagues from IOCB CAS characterized interactions between p53 and FOXO4 at the molecular level. New knowledge about the structure of the complex may enable the development of specific inhibitors of the interaction between these two proteins, and subsequently in the development of new drugs aimed at the selective elimination of senescent cells.
In this structural study, the researchers performed a detailed characterization of the interactions in the FOXO4: p53 complex using an integrated approach involving analytical ultracentrifugation, nuclear magnetic resonance, and chemical cross-linking coupled to mass spectrometry. Because both FOXO4 and p53 have multiple domains (see Figure), they studied the role of individual domains and disordered segments of both proteins and mapped their interaction interfaces. They found out that the interaction between p53 transactivation domain TAD and the FOXO4 Forkhead domain is crucial for the overall stability of the p53:FOXO4 complex. Furthermore, contacts involving the N-terminal disordered FOXO4 segment, the C-terminal negative regulatory domain of p53, and the DNA-binding domains of both proteins stabilize the complex formation. By measuring DNA binding, they further found that the p53: FOXO4 complex formation blocks p53 binding to DNA without affecting the DNA-binding properties of FOXO4.
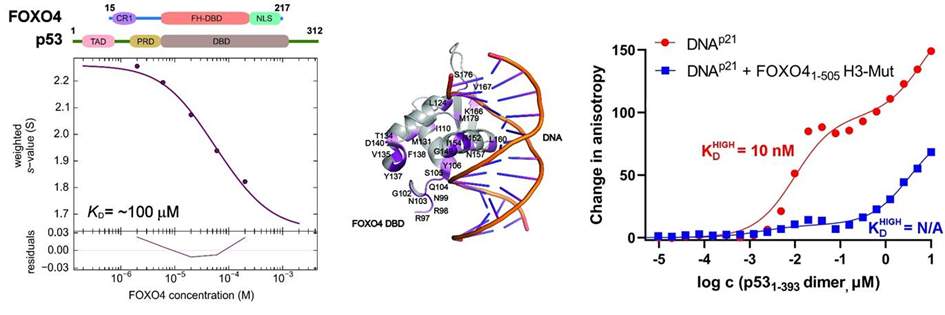
Left, sedimentation velocity analytical ultracentrifugation analysis of interaction between FOXO4 and p53. Middle, chemical shift perturbations obtained from 1H-15N HSQC spectra of 15N-labeled FOXO4 in the presence of p53 mapped onto the crystal structure of the FOXO4 DBD:DNA complex. Right, fluorescence anisotropy measurements showing that the complex formation reduces the DNA-binding affinity of p53.
Mandal R, Kohoutova K, Petrvalska O, Horvath M, Srb P, Veverka V, Obsilova V and Obsil T. FOXO4 interacts with p53 TAD and CRD and inhibits its binding to DNA. Protein Sci. roč. 31, č. 5 (2022), č. článku e4287. IF = 6.725. DOI
NEW PUBLICATION
An oral glucose tolerance test (OGTT) is the most commonly used method to diagnose diabetes mellitus from a drop of blood. It measures the ability of an organism to clear circulating glucose after ingestion of glucose bolus after an overnight fast. Although the dynamics of the blood glucose levels during the OGTT are well known, much less information about the metabolic changes in the target organs and the inter-organ communication are available. In our study, we investigated what is the fate of the sugar molecules in each organ and how it affects metabolic pathways in the body. Therefore, we performed the OGTT in mice using glucose with stable isotopic tracers (13C), analyzed 13C6-glucose tissue distribution and time profiles of metabolites and lipids across 12 organs and plasma. We found, that during the OGTT, the glucose use is turned on with specific kinetics at the organ level, but fasting substrates like β-hydroxybutyrate are switched off in all organs simultaneously. Timeline profiling of 13C-labeled fatty acids and triacylglycerols across tissues suggests that brown adipose tissue may contribute to the circulating fatty acid pool at maximal plasma glucose levels. We have created a virtual interactive atlas of metabolites (sugars, amino acids, lipids, etc.), which describes the interactions between organs after ingesting grape sugar. Metabolic fate of ingested glucose carbons was followed in 12 organs and plasma.

Visit the web application to explore virtual mouse metabolome yourself.
Lopes M, Brejchova K, Riecan M, Novakova M, Rossmeisl M, Cajka T, Kuda O. Metabolomics atlas of oral 13C-glucose tolerance test in mice. Cell Rep. 2021 Oct 12;37(2):109833. DOI. PMID: 34644567 IF = 9,423
The effectivity of opioid analgesics is reduced by CCL2 chemokine (8.12. 2021)
Opioid analgesics are the standard of care in the treatment of serious painful states. Treatment of neuropathic pain states, induced by damage to the nervous system, is especially difficult and opioid analgesics often do not have a beneficial effect. It was shown before that neuropathic states are accompanied with neuroinflammatory changes in the spinal cord and the level of different signaling molecules such as chemokine CCL2 is increased. This work shows that chemokine CCL2 is one of the important factors significantly reducing the effectivity of analgesics acting through opioid receptors. It acts probably directly on neurons and also through activation of microglial cells. Analgesic treatment with opioids has also number of serious unwanted side effects. One of them is a paradoxical increase of sensitivity, hyperalgesia/pain after opioids administration. This work shows that this hyperalgesia may be related to TRPV1 receptors activation. These published results suggest that to improve pain treatment with opioid analgesics, modulation of CCL2 and TRPV1 receptors may be needed, especially in cases of neuropathic pain.

The inhibition of the opioid agonists induced reduction of painful/nociceptive signaling in the spinal cord dorsal horn by the CCL2 chemokine. The control blue line demonstrates the inhibition of the nociceptive synaptic signaling after the DAMGO application and later (in about 13minutes) its potentiation. DAMGO is µ opioid receptors agonist and simulates thus application of opioid analgesics. The analgesic effect of DAMGO application is completely reversed in the presence of CCL2 chemokine (red line). The purple line demonstrates that the effect of CCL2 is dependent on microglia cells activation as it is prevented by microglia inhibitor minocycline.
Chemokine CCL2 preventsopioid‑inducedinhibitionofnociceptivesynaptictransmission in spinalcorddorsalhorn. Mario Heles, Petra Mrozkova, Dominika Sulcova, Pavel Adamek, Diana Spicarova and Jiri Palecek, JournalofNeuroinflammation (2021) 18:279, DOI, IF=8.23
Laboratory of Pain Research, Institute of Physiology, The Czech Academy of Sciences, Videnska 1083, 142 20 Praha 4, Czech Republic
Gliflozins - more than just antidiabetics (8.12. 2021)
Gliflozins are commonly prescribed for the treatment of diabetes (patients may know e.g. empagliflozin under the brand name Jardiance). Gliflozins inhibit the activity of sodium-glucose transporter in kidneys, which leads to higher glucose excretion in urine and normalization of blood glucose levels. Besides alleviation of hyperglycemia, other beneficial effects were observed in patients taking gliflozins, including decreased body weight, reduced blood pressure or improved kidney function. How this is possible? Here comes the right time to take a step backwards and to look once again and in greater detail on gliflozins effects in rodent models. And that is exactly what dr. Vaneckova and her colleagues from IKEM and departments of IPHYS are doing. They characterized the effects of empagliflozin in various non-diabetic rodent models prone to hypertension (Ren-2 transgenic rats; TGR) and lipid imbalance (hereditary hypertensive rats). Although the details of treatment effects vary among the individual models, empagliflozin generally attenuated inflammation, and normalized plasma and tissue lipid levels. In addition, it inhibited the activity of sympathetic nervous system, which resulted in a decrease of blood pressure in TGR. Hopefully, this and similar findings will allow to extend the use of gliflozins in the future to more diagnoses than just diabetes.
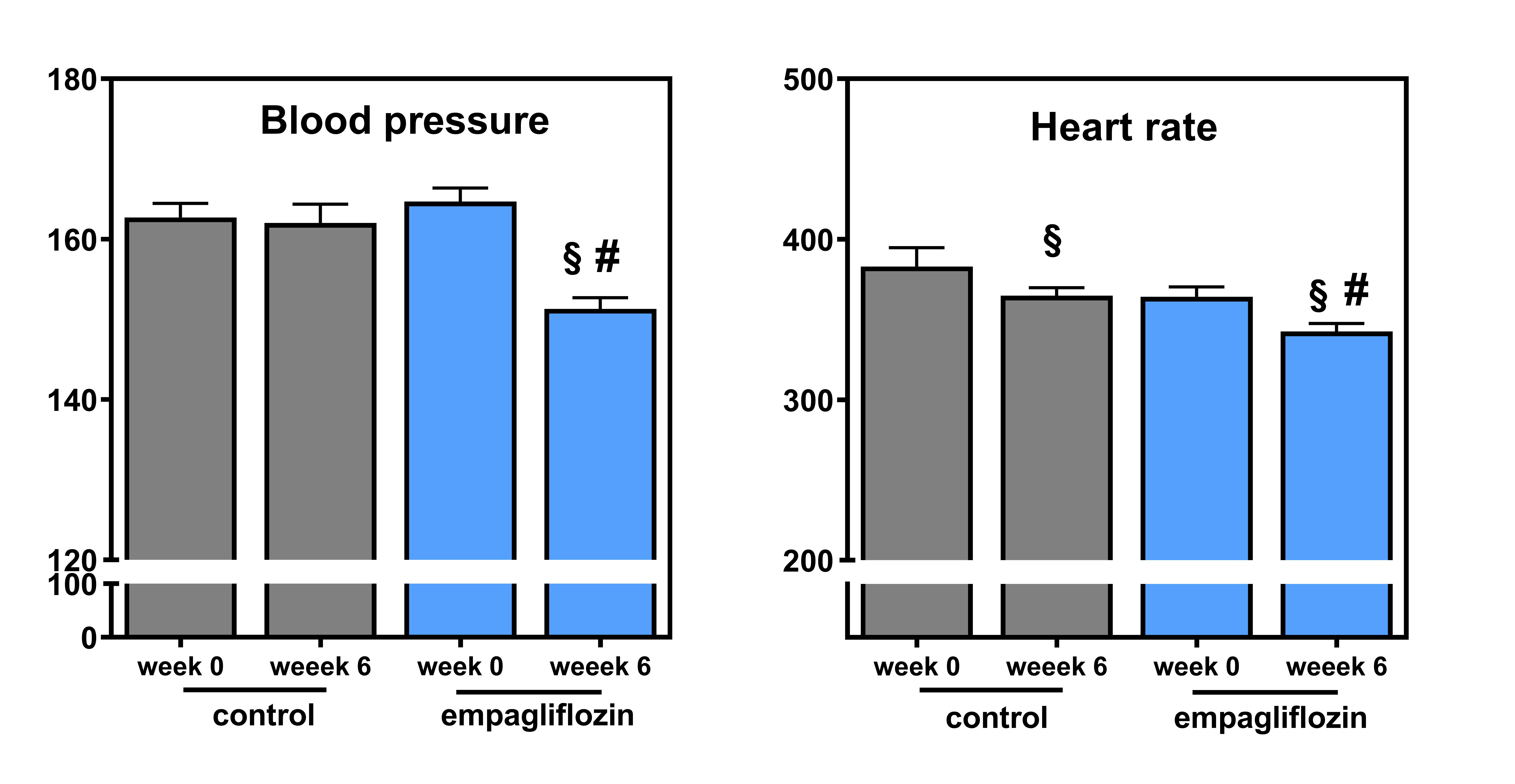
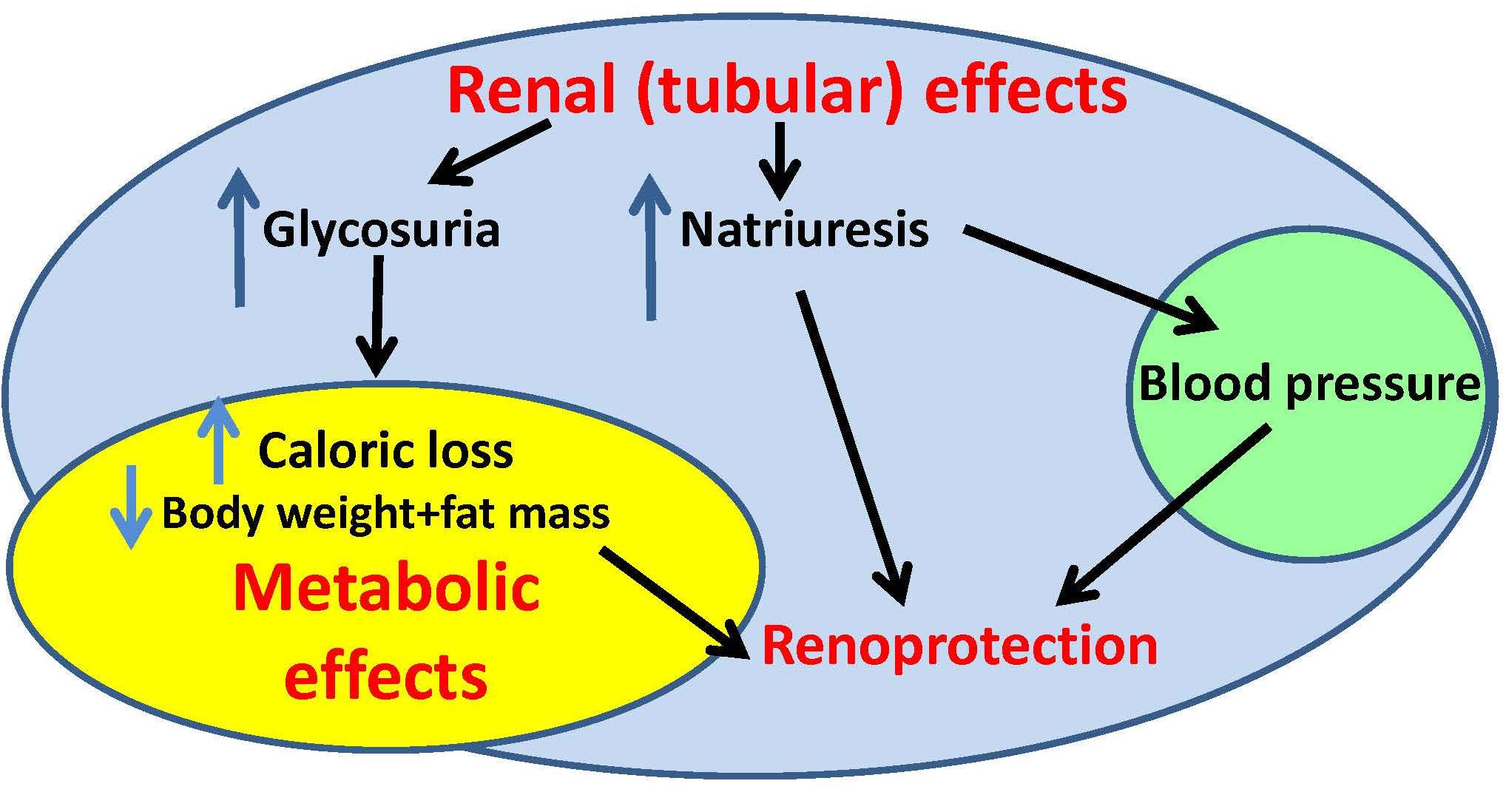
Hojná S, Rauchová H, Malínská H, Marková I, Hüttl M, Papoušek F, Behuliak M, Miklánková D, Vaňourková Z, Neckář J, Kadlecová M, Kujal P, Zicha J, Vaněčková I. Antihypertensive and metabolic effects of empagliflozin in Ren-2 transgenic rats, an experimental non-diabetic model of hypertension. Biomed Pharmacother. 2021 Dec;144:112246. Epub 2021 Oct 1. IF-5,98, DOI
Load next






